|
The surface levels of CD163 and CD206 were reported to be reduced in accutase-treated macrophages 1. Accutase is considered a mild-acting enzyme and does not affect most cell surface markers, including CD14, CD117, CD49f and CD29 2, 5. Trypsin cleaves peptides after lysine or arginine residues that are not followed by proline and degrades most cell surface proteins depending on the incubation time. The enzymes trypsin and accutase (Accutase ®, Innovative Cell Technologies) are frequently used for detaching adherent cells. The loss of cell surface proteins due to excessive enzymatic degradation may influence the analysis of surface markers and reduce cell viability, especially that of stem cells 4. However, most surface proteins and extracellular matrix components are degraded during enzymatic digestion. Proteolytic enzyme digestion is a frequently used method for cellular detachment. However, it is usually not sufficiently potent for strongly adherent cells and requires mechanical dislodgement by scraping, which may inadvertently tear the cells. EDTA is a mild method of cell detachment. The calcium chelator ethylenediaminetetraacetic acid (EDTA) removes calcium ions that are required for integrins to maintain cell adhesion. Some strongly adherent cells require other means to break the interactions between cell proteins and the surface of the dish, thus allowing detachment from the dish. Some lightly adherent cells can be lifted from the dish simply by spraying with phosphate buffered saline and tapping the dish. Therefore, efficient and reliable detachment methods are critical for studying the biological characteristics of cultured adherent cells. 
Cell detachment methods are also important for the stem cell maintenance of stemness 2, 3. When culturing certain cells, such as macrophages, which may adhere strongly to culture dishes, certain cell detachment strategies are required to achieve effective separation while simultaneously maintaining cell survival. However, the method of cell detachment may influence the surface proteins and phenotypes of cells cultured in vitro 1. A variety of harvesting techniques have been employed to detach cultured adherent cells in vitro for functional and phenotypic analyses. In the culture of adherent cells, cell detachment strategies are essential for cell harvest and are usually facilitated by mechanical methods combined with a calcium chelator or enzymatic cleavage of adhesion proteins to achieve effective detachment while maintaining cell viability. Traditionally, adherent cells are cultured in monolayers adherent on flat culture plates or dishes and require detachment methods to release them before further passage or experiments. These findings indicate that it is important to choose suitable cell detachment buffers and allow cells to recover after detachment before experiments. We demonstrated that using accutase to dissociate adherent cells compromised the expression of Fas ligands and Fas receptors on the cell surface. Our results also illustrated that surface proteins required 20 h to recover after accutase treatment. Moreover, we found that accutase may be able to cleave surface Fas ligands into pieces. Compared with ethylenediaminetetraacetic acid (EDTA)-based nonenzymatic cell dissociation buffers, accutase was associated with significant decreases in the surface Fas ligands and Fas receptors. In this study, we demonstrated that use of accutase for cellular detachment may compromise some surface proteins. A mild cell detachment buffer, accutase, is recommended for the replacement of trypsin to dissociate adherent cells and thereby avoid cellular damage. 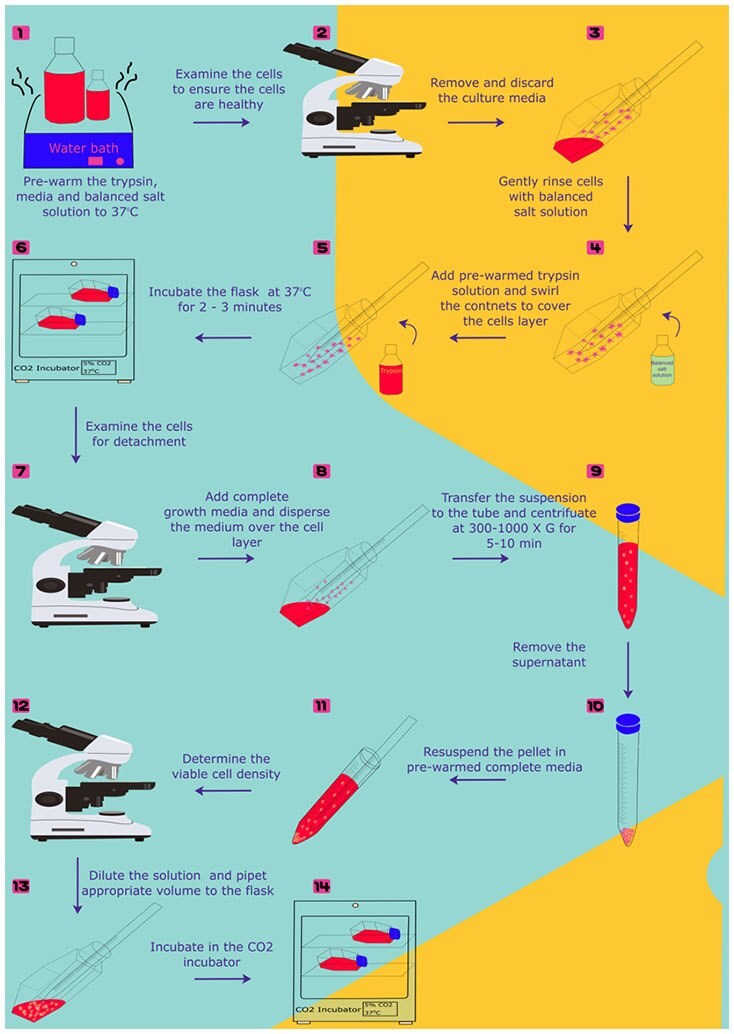
However, most surface proteins and the extracellular matrix are degraded by enzymatic digestion. Trypsinization is frequently used for cellular dissociation and detachment. Therefore, it is important to harvest cells with a proper detaching method to maintain the viability of cells after detachment. In cell culture environment, some cells adhere firmly to the culture plates and may be vulnerable to cell detachment during passage.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
